Research
Peptide Research and Gut Inflammatory Disorders
Irritable Bowel Syndrome (IBS)
Irritable bowel syndrome (IBS) is a chronic disorder of the gastrointestinal tract, characterized by abdominal pain and alterations in bowel habits. IBS is the disorder most commonly encountered by gastroenterologists, and diagnosis is made according to a symptom-based classification system, the Rome Criteria, with the latest version, Rome IV, recently released (Drossman, 2016). Prevalence rates in North America have been reported as approximately 12%, and symptoms occur more often in patients less than 50 years of age.
Patients with IBS suffer not only from gastrointestinal distress, but approximately 40–60% experience comorbid psychological disorders, such as depression or anxiety. In addition, patients with IBS report higher levels of somatization compared to patients without IBS but with gastrointestinal symptoms. Not surprisingly, IBS has been shown to negatively impact patients’ quality of life, as well as to adversely affect society’s financial resources.
Despite the substantial cost of IBS to both patients and society, curative, medical interventions have yet to be discovered. The development and persistence of IBS symptoms have been acknowledged as multifactorial in nature, making treatment of the disorder a complicated, clinical endeavor. Approaches are based on the reduction of patient symptomatology, and current pharmacological management often provides suboptimal relief.
Evidence of biological dysregulation has been reported in patients with IBS, and efforts are ongoing to advance understanding of the neurohormonal underpinnings of the disorder. Although gains have been made in understanding the pathophysiology of IBS, exact mechanisms leading to symptom development are not completely understood.
Inflammatory bowel disease (IBD)
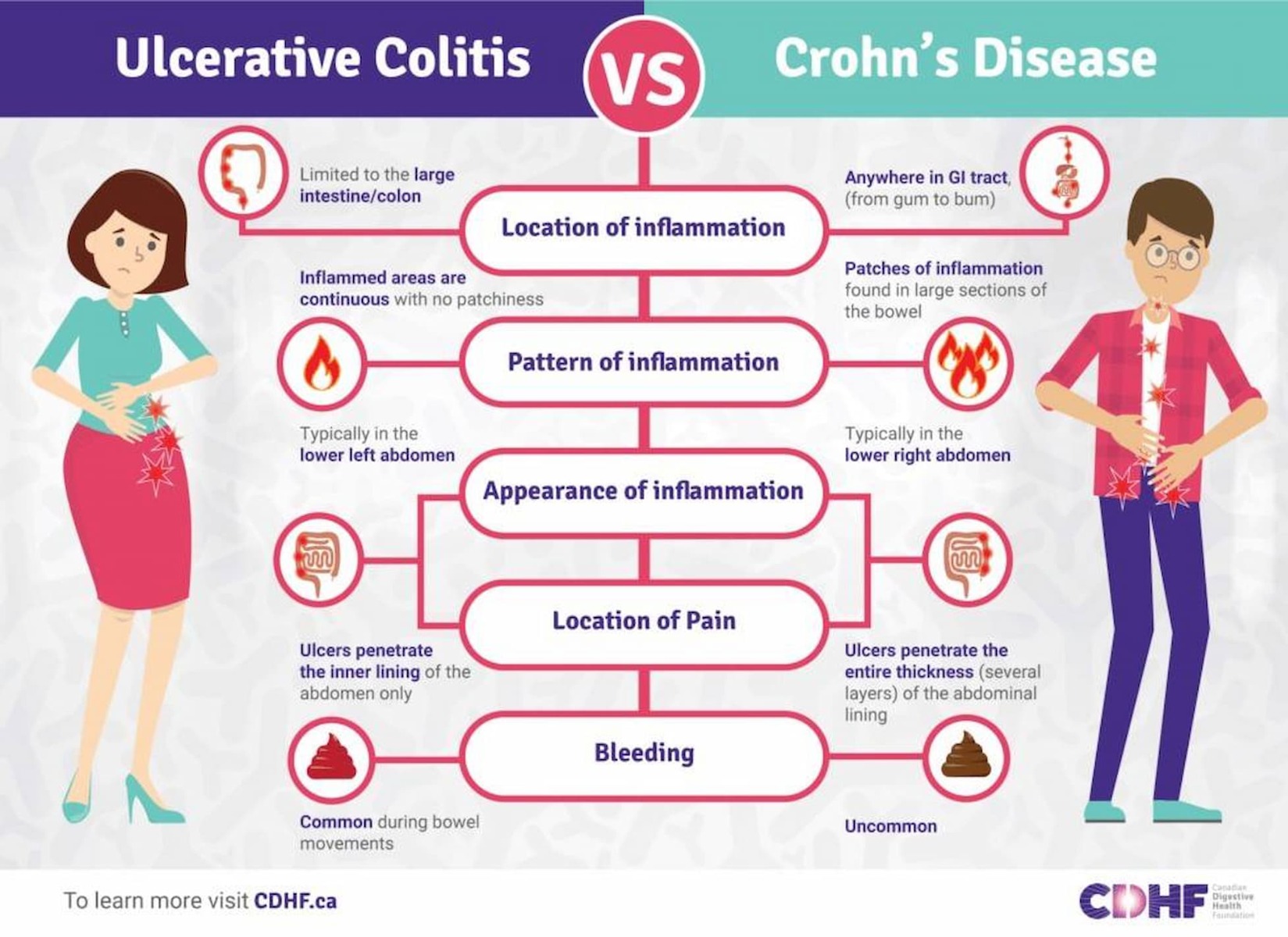
The inflammatory bowel diseases primarily include Crohn’s disease and ulcerative colitis. Crohn’s disease is an IBD that causes inflammation anywhere along the lining of the digestive tract, while ulcerative colitis causes long-lasting inflammation in some part of the digestive tract (mainly the colon). The exact etiology of IBD is not well known.
There are several factors that have been postulated to influence the development of this group of diseases, which include but are not limited to bacterial contamination, a change in the immune system, and genetic variations. For instance, a mutation in the NOD2 gene is associated with an increased susceptibility to IBD via production of proinflammatory cytokines. While genetic predisposition plays a key role in immune-mediated diseases, the major influence appears to be due to environmental factors.
Current research suggests that autoimmune diseases are most prevalent in highly industrialized nations but rare in less developed countries. Moreover, studies have shown that increased consumption of milk protein, animal protein, and polyunsaturated fatty acids can increase the risk for IBD, and that consumption of tobacco increases the risk of Crohn’s disease.
The major subtypes of IBD, including Crohn’s disease and ulcerative colitis, have a high prevalence rate in the world, with North America noting the highest frequency of people suffering with Crohn’s disease. In addition, statistics show that an estimated 129,000 people live with the disease in Canada. Although the onset of the disease usually occurs during adulthood, children are increasingly being diagnosed with IBD.
Treating IBD often involves use of medications that can diminish the symptoms and decrease the inflammation in the colon lining. A group of anti-inflammatory drugs including 5-aminosalicylic acid is commonly used to treat IBD. Other drugs such as infliximab are also indicated in patients who have failed conventional therapy and are hospitalized with severe IBD. Infliximab is a chimeric monoclonal antibody against tumor necrosis factor alpha (TNF-α), a cytokine involved in intestinal inflammation.
Several other immunomodulatory drugs, such as thalidomide, can also be used to treat a patient with severe IBD. Formerly used as a sedative and hypnotic, this synthetic drug has been shown to significantly reduce the inflammation associated with IBD. However, under certain circumstances, when medical therapy fails, surgery may be considered. This operation is known as colectomy and involves removal of the large intestine. While ulcerative colitis is cured upon removal of the colon, Crohn’s disease unfortunately can still recur after surgery.
While medication is commonly used to treat IBD, most pharmaceutical compounds have side effects such as headache, diarrhea, and nausea, which can reduce patient compliance and result in worsening of the condition. Therefore, different approaches to therapy like the use of peptides, can provide promising results for those who have gut inflammatory disorders.
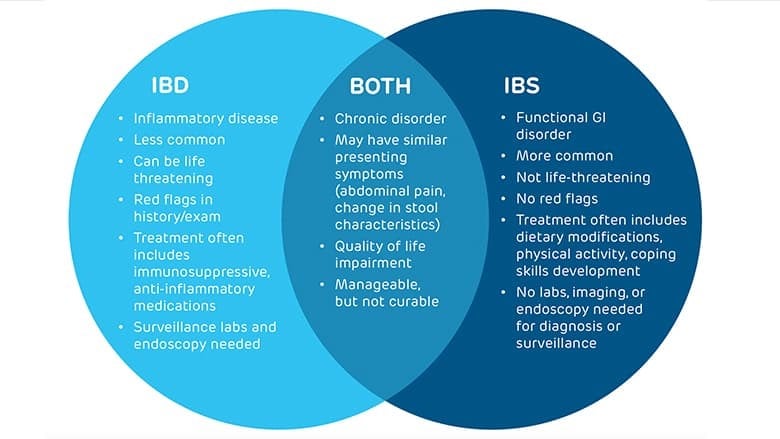
What is the difference between IBD and IBS?
Inherently, IBD is an organic disease, as evidenced by mucosal inflammation, whereas IBS lies more in the spectrum of a functional disorder, with no evidence of organic disease. IBS symptoms are nonspecific and may precede diagnosis of both IBS and IBD by many years.
See the Venn diagram below to understand the relationship among IBD and IBS.
What is KPV Peptide?
α-MSH is a neurohormone with extensive immunomodulatory effects on certain cell types. It exhibits potent anti-inflammatory effects by binding to melanocortin receptors in the skin. It is reported that the anti-inflammatory activity of α-MSH is mediated by the three terminal amino acids, Lysine-Proline-Valine (KPV).
KPV lacks the entire sequence motif required for binding to the MC-Rs, but still retains almost all the anti-inflammatory capacity of the parent hormone. This is also because peptides (like KPV) often act as hormones and relay information from one tissue through the blood to another via biologic messengers. The inflammatory activity of KPV is suggested to be mediated via inhibition of interleukin (IL)-1β.
Unlike α-MSH, KPV is free from the melanotropic effects and thus does not cause any pigmentation. Additionally, it is smaller in size and more chemically stable than α-MSH. These attributes make KPV a suitable candidate for the treatment of inflammatory skin disorders. KPV has a molecular weight of 383.49 Da and an Isoelectric point (pI) of 14 and hydrophilic in nature.
KPV is more stable and exerts less side effects, as it shares its sequence with human proteins. KPV might therefore be an interesting, easy to produce, and inexpensive therapeutic option in the treatment of inflammatory bowel disease that must be elucidated in future clinical trials.
Research Example #1:
Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease
Background: Despite some progress in recent years, the options for treating inflammatory bowel disease (IBD) are still dissatisfying, and surgery rates are still high. The anti-inflammatory effects of melanocortin peptides such as alpha-melanocyte-stimulating hormone (alpha-MSH) have been described recently in, for example, dextran sodium sulfate (DSS) colitis in mice. The aim of this study was to investigate the therapeutic potential of the melanocortin-derived tripeptide alpha-MSH(11-13) (KPV) and its mode of action in 2 models of intestinal inflammation.
Methods: The anti-inflammatory activity of KPV was analyzed in 2 well-described models of IBD: DSS colitis, and CD45RB(hi) transfer colitis. Furthermore, animals expressing a nonfunctional melanocortin-1 receptor (MC1Re/e) received DSS for induction of colitis and were treated with KPV. The course of inflammation was monitored by weight loss and histological changes in the colon as well as by myeloperoxidase (MPO) activity.
Results: In the DSS-colitis model, treatment with KPV led to earlier recovery and significantly stronger regain of body weight. Histologically, inflammatory infiltrates were significantly reduced in KPV-treated mice, which was confirmed by the significant reduction of MPO activity in colonic tissue after KPV treatment. Supporting these findings, KPV treatment of transfer colitis led to recovery, regain of body weight, and reduced inflammatory changes histologically. In MC1Re/e mice, KPV treatment rescued all animals in the treatment group from death during DSS colitis.
Conclusions: The melanocortin-derived tripeptide KPV showed significant anti-inflammatory effects in 2 murine models of colitis. These effects seem to be at least partially independent of MC1R signaling. In conclusion, our data suggest KPV as an interesting therapeutic option for the treatment of IBD.
Research Example #2:
PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation
Background & aims: KPV is a tripeptide (Lys-Pro-Val), which possesses anti-inflammatory properties; however, its mechanisms of action remain unknown. PepT1 is a di/tripeptide transporter normally expressed in the small intestine and induced in colon during inflammatory bowel disease (IBD). The aim of this study was to 1) investigate whether the KPV anti-inflammatory effect is PepT1-mediated in intestinal epithelial and immune cells, and 2) examine the anti-inflammatory effects in two models of mice colitis.
Methods: Human intestinal epithelial cells Caco2-BBE, HT29-Cl.19A, and human T cells (Jurkat) were stimulated with pro-inflammatory cytokines in the present or absence of KPV. KPV anti-inflammatory effect was assessed using a NF-kappaB luciferase gene reporter, Western blot, real-time RT-PCR and ELISA. Uptake experiments were performed using cold KPV as a competitor for PepT1 radiolabeled substrate or using [(3)H] KPV to determine kinetic characteristics of KPV uptake. Anti-inflammatory effect of KPV was also investigated in DSS- and TNBS-induced colitis in mice. KPV was added to drinking water and inflammation was assessed at the histologic level and by proinflammatory cytokine mRNA expression.
Results: Nanomolar concentrations of KPV inhibit the activation of NF-kappaB and MAP kinase inflammatory signaling pathways and reduce pro-inflammatory cytokine secretion. We found that KPV acts via PepT1 expressed in immune and intestinal epithelial cells. Furthermore, oral administration of KPV reduces the incidence of DSS- and TNBS-induced colitis indicated by a decrease in pro-inflammatory cytokine expression.
Conclusions: This study indicates that KPV is transported into cells by PepT1 and might be a new therapeutic agent for IBD.
What is BPC-157 Peptide?
BPC 157 is a penta-decapeptide containing a partial sequence of the body protection compound (BPC) isolated from the human gastric juice]. It is stable and resistant to hydrolysis or digestion by enzymes. Previous studies have demonstrated the promoting effect of BPC 157 on the healing of different tissues, including skin, mucosa, cornea, muscle, tendon, ligament and bone in animal studies. The mechanism by which penta-decapeptide BPC 157 accelerates healing is not clearly understood. It has been suggested to include up-regulation of growth factors, proangiogenic effect, and modulation of nitric oxide (NO) synthesis. BPC 157 may also control functions of collagen fragments that are associated with bone morphogenic proteins.
Research Example #1:
Stable gastric penta-decapeptide BPC 157 in trials for inflammatory bowel disease (PL-10, PLD-116, PL14736, Pliva, Croatia) heals ileoileal anastomosis in the rat
Purpose: Gastric penta-decapeptide BPC 157 (BPC 157), which has been shown to be safe in clinical trials for inflammatory bowel disease (PL-10, PLD-116, PL14736, Pliva, Croatia), may be able to cure intestinal anastomosis dehiscence. This antiulcer peptide shows no toxicity, is limit test negative, and a lethal dose is not achieved. It is stable in human gastric juice. In comparison with other standard treatments, it is more effective for ulcers and various wounds, and can be used without a carrier needed for other peptides, both locally and systemically (i.e., perorally, parenterally). We studied the effectiveness of BPC 157 for ileoileal anastomosis healing in rats.
Methods: We assessed ileoileal anastomosis dehiscence macroscopically, histologically, and biomechanically (volume [ml] infused through a syringe-perfusion pump system (1 ml/10 s), and pressure [mmHg] to leak induction [catheter connected to a chamber and a monitor, at 10 cm proximal to anastomosis]), at 1, 2, 3, 4, 5, 6, 7, and 14 days. BPC 157 (10 microg, 10 ng, 10 pg/kg i.p. (or saline [5 ml/kg]) was first administered after surgery, while it was last given 24 h before either assessment or sacrifice.
Results: Throughout the experiment, both higher doses of BPC 157 were shown to improve all parameters of anastomotic wound healing. The formation of adhesions remained slight, the blood vessels were filled with blood, and a mild intestinal passage obstruction was only temporarily observed. Anastomosis without leakage induces markedly higher volume and pressure values, with a continuous increase toward healthy values. From day 1, edema was markedly attenuated, and the number of granulocytes decreased, while from days 4 or 5 necrosis decreased and granulation tissue, reticulin, and collagen formation substantially increased, thus resulting in increased epithelization.
Conclusion: This study showed BPC 157 to have a beneficial effect on ileoileal anastomosis healing in the rat.
Research Example #2:
Penta-decapeptide BPC 157 positively affects both non-steroidal anti-inflammatory agent-induced gastrointestinal lesions and adjuvant arthritis in rats
Besides a superior protection of the penta-decapeptide BPC 157 (an essential fragment of an organ protective gastric juice peptide BPC) against different gastrointestinal and liver lesions, an acute anti-inflammatory and analgetic activity was also noted. Consequently, its effect on chronic inflammation lesions, such as adjuvant arthritis, and non-steroidal anti-inflammatory agents (NSAIAs)-induced gastrointestinal lesions was simultaneously studied in rats. In gastrointestinal lesions (indomethacin (30 mg/kg sc), aspirin (400 mg/kg ig) and diclofenac (125 mg/kg ip) studies, BPC 157 (10 μg or 10 ng/kg ip) was regularly given simultaneously and/or 1 h prior to drug application (indomethacin). In the adjuvant arthritis (tail-application of 0.2 mL of Freund’s adjuvant) studies (14 days, 30 days, 1 year) BPC 157 (10 μg or 10 ng/kg ip), it was given as a single application (at 1 h either before or following the application of Freund’s adjuvant) or in a once daily regimen (0–14th day, 14–30th day, 14th day–1 year). Given with the investigated NSAIAs, BPC 157 consistently reduced the otherwise prominent lesions in the stomach of the control rats, as well as the lesions in the small intestine in the indomethacin groups. In the adjuvant arthritis studies, the lesion’s development seems to be considerably reduced after single penta-decapeptide medication, and even more attenuated in rats daily treated with BPC 157. As a therapy of already established adjuvant arthritis, its salutary effect consistently appeared already after 2 weeks of medication, and it could be clearly seen also after 1 year of application. Taking together all these results, the data likely point to a special anti-inflammatory and mucosal integrity protective effect.
What is VIP Peptide?
Vasoactive intestinal peptide (VIP) is a 28-residue amino acid peptide first characterized in 1970 that was initially isolated from porcine duodenum. A member of the secretin/glucagon hormone superfamily VIP is evolutionarily well conserved with sequence similarity among fish, frogs, and humans; among mammals, except for guinea pigs and chickens, the sequence similarity is at least 85%.
VIP was initially discovered owing to its potent vasodilatory effects (as its name implies). VIP is widely distributed in the central and peripheral nervous system as well as in the digestive, respiratory, reproductive, and cardiovascular systems as a neurotransmitter and neuroendocrine releasing factor. These effects contribute to an extensive range of physiological and pathological processes related to development, growth, and the control of neuronal, epithelial, and endocrine cell function. VIP has also been implicated in the regulation of carcinogenesis, immune responses, and circadian rhythms.
How does VIP work?
The two receptors that recognize VIP, termed VPAC1 and VPAC2, are class B of G-protein-coupled receptors (GPCRs), also known as the secretin receptor family, which includes receptors for VIP, PACAP, secretin, glucagon, glucagon-like peptide (GLP)-1 and -2, calcitonin, gastric inhibitory peptide (GIP), corticotropin-releasing factor (CRF)-1 and -2, and parathyroid hormone (PTH). VPAC1 and VPAC2 are activated by VIP and PACAP, whereas PACAP has its own specific receptor, named PAC1, for which VIP has very low affinity. Through these receptors, VIP can mediate an extensive number of GI functions such as regulating gastric acid secretion, intestinal anion secretion, enzyme release from the pancreas, cellular motility, vasodilation, and intestinal contractility. The localization of VIP, VPAC1, and VPAC2 is closely related to their physiological and pathological functions.
Research Example #1:
Vasoactive intestinal peptide decreases inflammation and tight junction disruption in experimental necrotizing enterocolitis
Background and Purpose
Excessive inflammatory cell infiltration and accumulation in the intestinal mucosa are pathological features of necrotizing enterocolitis (NEC) leading to intestinal barrier disruption. Vasoactive intestinal peptide (VIP) is a potent anti-inflammatory agent that regulates intestinal epithelial barrier homeostasis. We previously demonstrated that VIP-ergic neuron expression is decreased in experimental NEC ileum, and this may be associated with inflammation and barrier compromise. We hypothesize that exogenous VIP administration has a beneficial effect in NEC.
Methods
NEC was induced in C57BL/6 mice by gavage feeding, hypoxia, and lipopolysaccharide administration between postnatal day (P) 5 and 9. There were four studied groups: Control (n = 6): Breast feeding without stress factors; Control + VIP (n = 5): Breast feeding + intraperitoneal VIP injection once a day from P5 to P9; NEC (n = 9): mice exposed to NEC induction; NEC + VIP (n = 9): NEC induction + intraperitoneal VIP injection. Terminal ileum was harvested on P9. NEC severity, intestinal inflammation, (IL-6 and TNFα), and Tight junctions (Claudin-3) were evaluated.
Results
NEC severity and intestinal inflammation were significantly decreased in NEC + VIP compared to NEC. Tight junction expression was significantly increased in NEC + VIP compared to NEC.
Conclusion
VIP administration has a beneficial therapeutic effect in NEC by reducing inflammation and tight junction disruption.
Research Example #2:
Recent advances in vasoactive intestinal peptide physiology and pathophysiology: focus on the gastrointestinal system
Vasoactive intestinal peptide (VIP), a gut peptide hormone originally reported as a vasodilator in 1970, has multiple physiological and pathological effects on development, growth, and the control of neuronal, epithelial, and endocrine cell functions that in turn regulate ion secretion, nutrient absorption, gut motility, glycemic control, carcinogenesis, immune responses, and circadian rhythms. Genetic ablation of this peptide and its receptors in mice also provides new insights into the contribution of VIP towards physiological signaling and the pathogenesis of related diseases. Here, the researchers discuss the impact of VIP on gastrointestinal function and diseases based on recent findings, also providing insight into its possible therapeutic application to diabetes, autoimmune diseases and cancer.
PHDP5 Research for Alzheimer Disease
Introduction
Currently, there is no cure for Alzheimer’s disease, a progressive neurodegenerative condition that affects millions of people worldwide. However, treatments are available to help manage its symptoms, such as memory loss, confusion, and difficulties with thinking and reasoning. While these therapies do not stop the disease, some have shown promise in slowing its progression, offering hope for improving the quality of life for those affected.
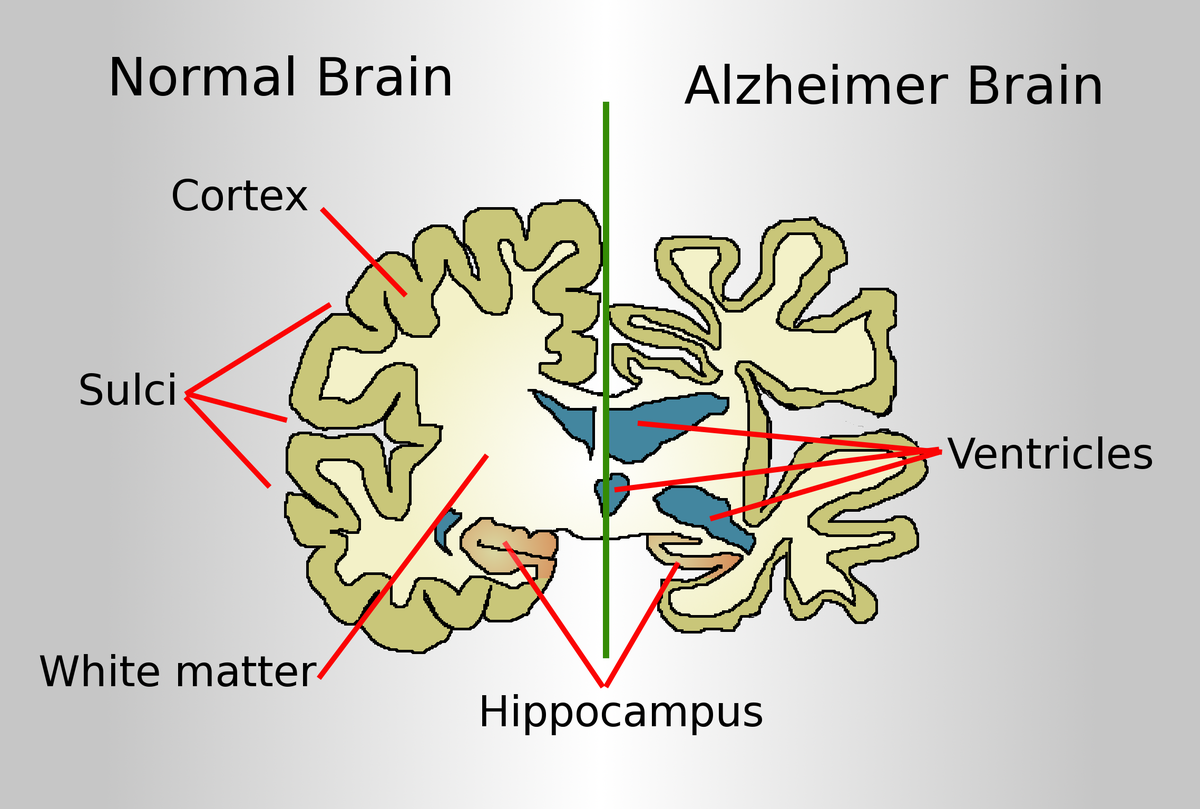
Alzheimer’s is characterized by the abnormal accumulation of proteins in the brain, such as beta-amyloid plaques and tau tangles, which interfere with the transmission of nerve impulses and disrupt normal brain function. These proteins build ups damage and ultimately kill brain cells, leading to the cognitive and functional decline associated with the disease.
In a groundbreaking study, researchers tested a synthetic peptide on mice genetically engineered to develop Alzheimer’s-like symptoms. The findings were promising, showing that the treatment significantly reduced the buildup of these harmful proteins in the brain. Even more compelling, the peptide therapy appeared to restore key cognitive functions, such as memory and learning, which are typically impaired in Alzheimer’s patients.
This study written about below highlights the potential of innovative therapeutic approaches to target the root causes of Alzheimer’s, rather than just addressing its symptoms. While further research, including human trials, is necessary to validate these findings, this development represents a significant step forward in the quest for more effective treatments and, ultimately, a cure for Alzheimer’s disease.
The problem
As global life expectancy continues to rise, dementia has become an increasingly pressing public health challenge. The prevalence of dementia is projected to grow significantly, with studies estimating that over 150 million people worldwide could be affected by 2050. This staggering statistic underscores the urgent need for effective prevention and treatment strategies.
Alzheimer’s disease, the most common form of dementia, is characterized by a variety of debilitating symptoms, including memory loss, cognitive decline, and personality changes. These symptoms are widely believed to result from the accumulation of two key proteins in the brain: beta-amyloid (Aβ) and tau. Beta-amyloid forms sticky plaques between neurons, while tau creates tangles within neurons, disrupting their structure and function. Together, these protein abnormalities lead to widespread neuronal damage, brain shrinkage, and the hallmark symptoms of Alzheimer’s.
Current treatments for Alzheimer’s primarily focus on alleviating symptoms, such as cognitive deficits and behavioral changes, rather than addressing the underlying causes of the disease. However, recent advancements have introduced disease-modifying therapies that aim to directly target these pathological proteins. For example, aducanumab and lecanemab, two monoclonal antibody treatments, have shown promise in reducing Aβ plaques in the brain. These therapies mark a significant step forward in targeting the disease at its root.
Despite these advancements, the use of monoclonal antibody treatments has sparked debate within the medical community. While they offer the potential to slow disease progression, their benefits are tempered by notable risks. Common side effects include brain swelling and microhemorrhages, raising concerns about the overall safety and efficacy of these treatments. Some experts question whether the modest clinical improvements observed in patients justify the associated risks, particularly considering the complex, multifactorial nature of Alzheimer’s disease.
As the global burden of dementia continues to rise, ongoing research is essential to refine these treatments, explore alternative therapeutic targets, and better understand the mechanisms underlying Alzheimer’s. The goal is to develop safer, more effective interventions that can not only slow disease progression but also prevent its onset, offering hope to millions of individuals and families affected by this devastating condition.
The possible solution
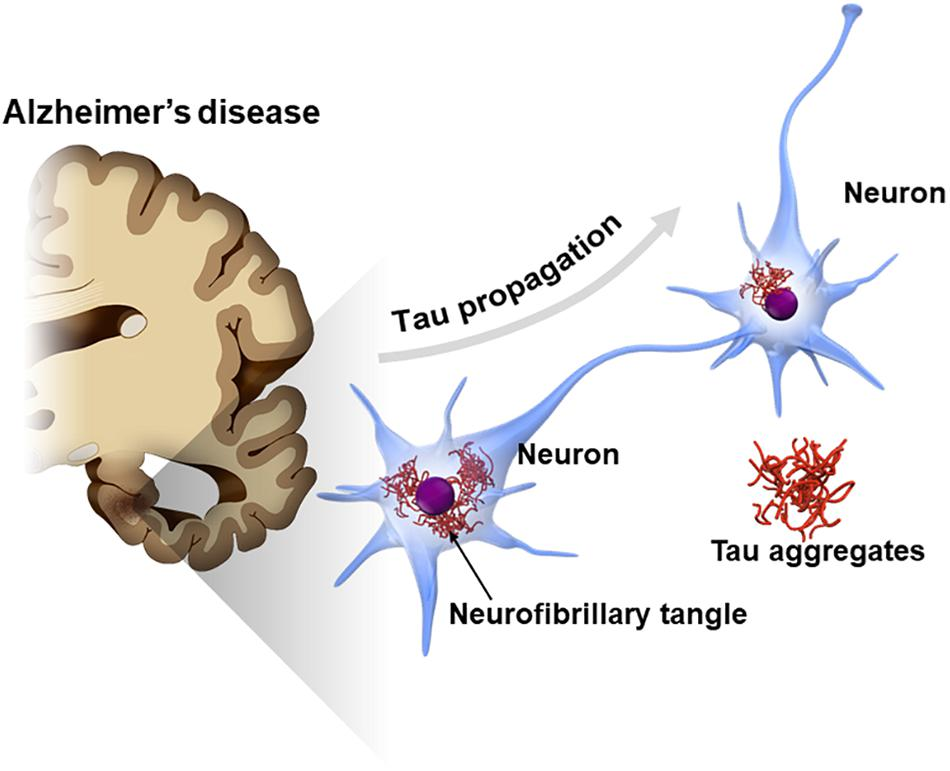
A recent study has introduced a promising new approach to treating Alzheimer’s disease by targeting tau protein accumulation. Tau proteins, when abnormally modified, form neurofibrillary tangles that disrupt the passage of nerve impulses across synapses, the critical junctions where nerve cells communicate. These tangles are a hallmark of Alzheimer’s and contribute significantly to the cognitive and memory impairments associated with the disease.
In this research, scientists tested the effects of a synthetic peptide, PHDP5, on transgenic mice engineered to develop Alzheimer’s-like symptoms. The peptide was found to inhibit a specific biochemical pathway responsible for tau buildup, effectively preventing the formation of these neurofibrillary tangles. Remarkably, the treatment not only reduced tau accumulation but also reversed deficits in memory and learning in the mice, suggesting its potential to restore cognitive functions impaired by Alzheimer’s.
The findings, published in the journal Brain Research, represent a significant step forward in understanding and addressing the complex mechanisms underlying Alzheimer’s disease. Unlike existing treatments that primarily target beta-amyloid plaques, this study highlights the importance of tau as a therapeutic target. By focusing on tau, the researchers offer a complementary approach that could work alongside beta-amyloid-targeting therapies to provide a more comprehensive strategy for tackling the disease.
Dr. Stefania Forner, the Alzheimer’s Association director of medical and scientific relations, who was not involved in the study, emphasized the significance of the findings. “Using a mouse model of Alzheimer’s disease, this study sheds some light on a novel potential treatment pathway,” she stated, underscoring the innovative nature of this research.
The use of PHDP5 as a therapeutic agent offers several advantages over traditional approaches. Targeting tau tangles directly could address a broader range of Alzheimer’s symptoms, particularly in patients where beta-amyloid-targeting therapies have shown limited efficacy. Additionally, the peptide’s ability to reverse memory and learning deficits provides hope for interventions that not only halt disease progression but also restore lost cognitive abilities.
Peptide reduces tau buildup in brain
The transmission of nerve impulses across synapses—the communication hubs between nerve cells—is a highly coordinated process that depends on two key components: dynamin and microtubules. These structures work in tandem to facilitate the recycling of vesicles, which are tiny sacs that store and release neurotransmitters. Neurotransmitters are essential chemicals that carry the nerve impulse from one neuron to another, ensuring seamless communication in the nervous system.
In healthy individuals, tau proteins play a critical role in stabilizing microtubules, which serve as the structural framework of nerve cells. This stability ensures that dynamin remains available to support vesicle recycling and maintain effective synaptic communication. However, in Alzheimer’s disease, tau proteins become dysfunctional. They detach from the microtubules, leading to their destabilization. This process also results in the removal of dynamin from nerve cells, disrupting the recycling of vesicles. Over time, the detached tau aggregates into tangles, a hallmark of Alzheimer’s disease, further impairing the transmission of nerve impulses and contributing to cognitive decline.
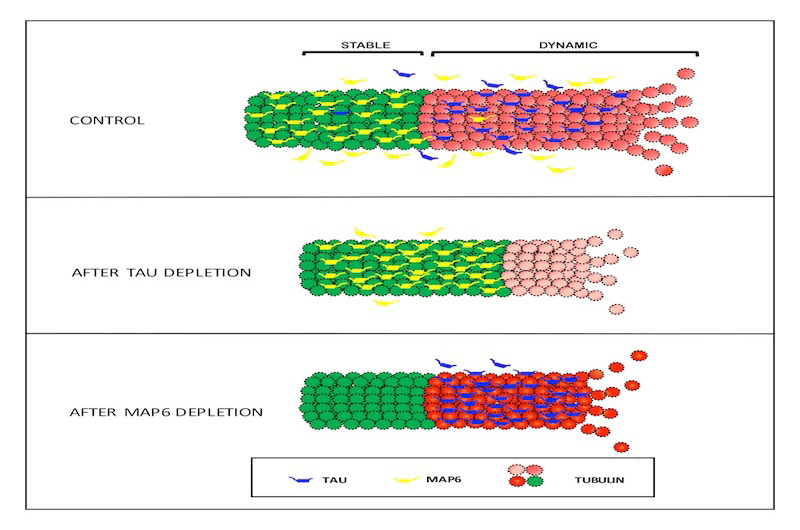
In earlier in vitro (laboratory-based) research, scientists explored the potential of a synthetic peptide, PHPD5, to counteract these damaging effects. The peptide was shown to release dynamin, making it available to support the recycling of vesicles and restore communication between synapses. By targeting this critical pathway, PHPD5 effectively bypassed the disruption caused by tau dysfunction, offering a novel mechanism to preserve synaptic health.
Building on these promising in vitro findings, recent studies have expanded to animal models, where PHPD5 demonstrated the ability to reverse memory and learning deficits by addressing tau-related disruptions. By stabilizing microtubules and freeing dynamin for vesicle recycling, the peptide restores a fundamental aspect of neuronal function that is severely compromised in Alzheimer’s disease.
This research examines the complexity of Alzheimer’s pathology and highlights the need for multifaceted therapeutic approaches. While much of the focus in Alzheimer’s treatment has traditionally been on targeting beta-amyloid plaques, these findings reinforce the critical role of tau in disease progression. By addressing tau-induced disruptions in synaptic communication, PHPD5 offers a complementary strategy that could work synergistically with beta-amyloid-targeting therapies.
Future research will need to investigate the long-term safety and efficacy of PHPD5 in human clinical trials. Additionally, understanding how this peptide interacts with other cellular mechanisms in the brain could pave the way for even more refined treatments. If successful, PHPD5 and similar therapies could not only halt the progression of Alzheimer’s but also restore lost neuronal functions, offering hope for reversing some of the devastating effects of this disease.
Dr. Emer MacSweeney, CEO and Consultant Neuroradiologist at Re:Cognition Health, who was not involved in the study, told Medical News Today: “This research appears to be pioneering in specifically targeting the dynamin-microtubule interaction with the synthetic peptide PHDP5. While other treatments for Alzheimer’s focus on different mechanisms, such as amyloid-beta plaques and tau tangles, targeting the dynamin-microtubule pathway is relatively novel. The researchers have shown in vitro and in vivo evidence of the positive effects of inhibiting this interaction, suggesting this could be the first time it has been targeted in this manner.”
Peptide treatment reaches the brain
Building on earlier findings that demonstrated the efficacy of the synthetic peptide PHPD5 in in vitro studies, researchers advanced their investigation by testing the peptide in a living model. They used transgenic Tau609 mice, a widely recognized model for Alzheimer’s disease research. These mice develop hallmark features of the disease, including tau tangles, neuronal loss, and memory deficits, making them an ideal subject for evaluating the therapeutic potential of PHPD5.
Study Design and Administration
The researchers administered 2 mg of PHPD5 in saline solution intranasally once daily for four weeks. This method was chosen for its ability to deliver therapeutics directly to the brain, bypassing systemic metabolism and efficiently crossing the blood-brain barrier. As a control, another group of Tau609 mice received only the saline solution via the same delivery method.
Cognitive Testing Using the Morris Water Maze (MWM)
To assess the impact of the treatment on memory and learning, the researchers employed the Morris Water Maze (MWM) test, a gold standard for evaluating spatial memory in animal studies. After three weeks of treatment, the mice underwent four training sessions in a circular water trough, 100 cm in diameter and 30 cm deep. The objective was for the mice to locate an escape platform submerged in one quadrant of the trough. This training phase was designed to test their ability to learn and remember the platform’s location.
Following the training sessions, the researchers conducted a memory test in the same water trough, but this time without the escape platform. They measured how much time the mice spent in the quadrant where the platform had been previously located. This provided a clear indication of the mice’s ability to recall the platform’s location, reflecting their spatial memory.
Results and Brain Analysis
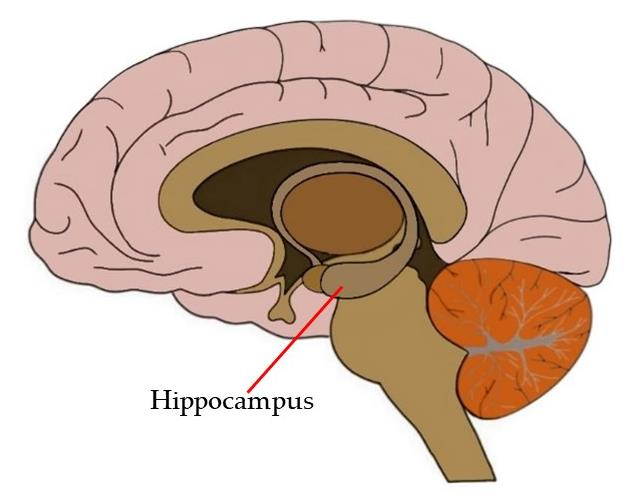
After the behavioral testing, the mice were euthanized, and their brains were examined to evaluate the physiological effects of PHPD5. Notably, the researchers found that the peptide successfully crossed the blood-brain barrier and accumulated in the hippocampus, the brain region critical for learning and memory. This finding is significant, as the hippocampus is particularly vulnerable to damage in Alzheimer’s disease, and effective therapeutic delivery to this region is a critical hurdle in neurodegenerative research.
Implications and Future Directions
The results of this study are highly encouraging. The successful delivery of PHPD5 to the hippocampus and its potential to restore learning and memory in Tau609 mice suggest that this peptide could offer a novel therapeutic avenue for treating Alzheimer’s disease and other tau-related neurodegenerative conditions. The intranasal administration route is particularly noteworthy, as it provides a non-invasive and efficient method of drug delivery to the brain. Future research will need to address several key questions. While the results in mice are promising, human trials are necessary to confirm the peptide’s safety, efficacy, and long-term effects. Additionally, researchers must explore the mechanisms by which PHPD5 interacts with tau pathology, as well as its potential impacts on other brain regions affected by Alzheimer’s disease.
If these findings translate to human patients, PHPD5 could represent a groundbreaking advancement in Alzheimer’s treatment—offering hope for not only slowing disease progression but also reversing some of its cognitive and neurological damage. This study exemplifies the importance of innovative research in tackling the complexities of neurodegenerative diseases.
Improvements in learning and memory
The researchers evaluated the effects of PHPD5 treatment on learning and memory by comparing the performance of three groups of mice in the Morris Water Maze (MWM): wild-type (WT) mice, untreated control Tau609 mice, and PHPD5-treated Tau609 mice. This comparison provided a comprehensive assessment of the peptide’s ability to restore cognitive functions impaired by tau pathology.
During the four-day training phase, the WT mice performed the best, demonstrating a 60% reduction in the time required to locate the hidden platform by the fourth session, reflecting their intact learning and memory capabilities. In contrast, the control Tau609 mice showed impaired learning, achieving only a 33% reduction in finding time over the same period, highlighting the cognitive deficits associated with tau accumulation. Remarkably, the PHPD5-treated Tau609 mice displayed significant improvements, with their performance closely mirroring that of the WT mice. By the fourth training session, the treated mice achieved a 55% reduction in the time needed to locate the platform, indicating substantial recovery of learning abilities.
On the fifth day, the researchers assessed memory retention by removing the platform and measuring how much time each group spent in the quadrant where the platform had been located. WT mice spent 36% of their time in the platform’s previous location, demonstrating strong spatial memory retention. PHPD5-treated Tau609 mice spent 33% of their time in the same quadrant, showing near-normal memory recall comparable to the WT group. In contrast, control Tau609 mice spent only 25% of their time in the platform quadrant, underscoring their impaired memory retention. These findings suggest that PHPD5 treatment effectively restored the learning and memory abilities of Tau609 mice to levels like those of healthy WT mice. The treated mice’s ability to improve their performance during training and retain the memory of the platform’s location highlights the peptide’s potential in addressing cognitive deficits caused by tau pathology.
These results align with previous observations that PHPD5 stabilizes tau and facilitates dynamin availability for vesicle recycling, thereby restoring synaptic communication. By mitigating the effects of tau tangles and neuronal dysfunction, the peptide appears to directly address the mechanisms underlying learning and memory impairments. The success of PHPD5 in this experimental setup has far-reaching implications for Alzheimer’s disease research and treatment development.
Future studies should focus on investigating the long-term effects of sustained treatment with PHPD5, conducting human trials to evaluate its safety and efficacy, and exploring its potential as part of combination therapies targeting both tau and beta-amyloid. Additionally, researchers should assess whether PHPD5 could benefit other tau-related neurodegenerative diseases, such as frontotemporal dementia or progressive supranuclear palsy. If the success seen in animal models translates to humans, PHPD5 could represent a breakthrough in Alzheimer’s treatment, offering hope for millions affected by the disease.
A new target for Alzheimer’s treatment?
These are early findings in a new target area for Alzheimer’s treatment, but the study has shown that, in mice, drugs administered intranasally can cross the blood-brain barrier to reach the part of the brain most affected by Alzheimer’s. It has also shown that the synthetic peptide can reverse some of the damage caused by tau in mice genetically engineered to develop Alzheimer’s-like pathophysiology. The research on PHDP5 and its potential to reverse cognitive decline in Alzheimer’s is very encouraging and represents a significant advancement in the search for effective treatments. This could pave the way for new treatments that might prevent or significantly delay the progression of cognitive symptoms in Alzheimer’s.
Dihexa: A Peptide Modulator of Synaptic Plasticity and Neural Regeneration
Simplified Summary
Dihexa is a small, lab-designed protein fragment (peptide) created for scientific research into brain repair, learning, and memory. In simple terms, Dihexa acts like a molecular “boost” for neural connections. It’s synthetic, meaning it isn’t found naturally in the body – instead, scientists engineered it based on a short hormone fragment (angiotensin IV) to better penetrate the brain and last longer. Early laboratory studies in cells and animals suggest Dihexa can enhance synaptic plasticity, which is the brain’s ability to form and reorganize connections between neurons. For example, experiments indicate Dihexa might increase the number of synapses (the communication links between brain cells) and encourage the growth of new neuronal branches, potentially by turning up certain growth signals in the brain. It appears to influence neurotrophic pathways – in particular, it partners with a natural brain growth factor called HGF to activate the c-Met receptor, a pathway involved in cell growth and survival. By doing so, Dihexa has shown effects like improved learning in memory-impaired rats and more robust neural networks in tissue studies. Importantly, all these findings come from preclinical research (laboratory and animal studies) only. Dihexa is strictly an experimental compound not approved or intended for human use. Scientists study it to understand how tiny peptides can potentially rebuild neural connections and support brain regeneration, helping us learn how the brain might maintain cognitive resilience.
Introduction
Peptide bioregulators and synthetic neuroactive peptides are a growing frontier in biomedicine. These are very short chains of amino acids (the building blocks of proteins) that can send biological signals to cells. Natural peptide bioregulators (like those from the thymus or pineal gland) have been studied for their ability to modulate organ function and gene activity in specific tissues. Dihexa represents a new twist on this concept: it is a hexapeptide derivative of a brain hormone fragment, deliberately engineered by scientists to probe mechanisms of neuroplasticity and brain repair. First identified by researchers at Washington State University, Dihexa originated from efforts to improve on angiotensin IV, a naturally occurring peptide hormone fragment that had hinted at cognitive effects. By modifying Ang IV’s structure to be more brain-accessible and long-lasting, scientists created Dihexa as a tool to enhance synaptic connectivity and cognitive processes in preclinical models. In other words, Dihexa is designed to mimic and amplify the brain’s own growth signals, providing a window into how boosting certain pathways might improve learning and memory.
The rationale behind developing small peptide analogs like Dihexa comes from a desire to study neuroplasticity and neuronal repair at the molecular level. Traditional neurological drugs often target neurotransmitters or symptoms, but peptides like Dihexa work upstream, nudging the cell’s own growth and survival programs. By creating an analog of a natural signaling molecule (Ang IV), researchers aimed to trigger pathways involved in memory formation, synapse development, and neuron regeneration more powerfully than the original hormone fragment could. Dihexa specifically was designed to engage the hepatocyte growth factor (HGF)/c-Met system – a pathway known for its role in cell growth and healing – to see how activating this system affects brain cells. It is important to note that Dihexa remains only a research compound: all investigations so far have been limited to cell cultures and animal models. Scientists utilize Dihexa in the laboratory to explore HGF/c-Met pathway modulation and to deepen our understanding of neuronal communication, synaptic plasticity, and the brain’s capacity for self-repair.
Molecular Origin & Structural Characteristics
Dihexa’s chemical identity is that of a modified hexapeptide derived from angiotensin IV. In chemical terms, it is often described as N-hexanoic-Tyr-Ile-(6)HomoPhe-His-Leu (with the full name being N-(1-oxohexyl)-Tyrosyl-(6-aminohexanoic)-Isoleucinamide). This jargon means that the peptide’s backbone is based on a short chain of six amino acids, but with strategic modifications: a fatty acid chain (hexanoic acid) is attached to one end, and one of the amino acids is a special variant (homophenylalanine) not found in the original hormone sequence. These tweaks were intentional – by adding the hexanoyl group and using an amino acid analog, the molecule became more lipophilic (fat-loving) and less prone to degradation by enzymes. The result is a peptide that can survive longer in the bloodstream and cross the blood–brain barrier more effectively than Ang IV itself, which is rapidly broken down and normally doesn’t enter the brain well. Dihexa’s small size (molecular weight on the order of only a few hundred daltons, ~500 Da) and these lipophilic modifications allow it to slip through biological membranes and reach brain tissue in animal studies.
One of the most notable structural features of Dihexa is its ability to bind with high affinity to hepatocyte growth factor (HGF). HGF is a large protein growth factor, and its receptor is a protein on cell surfaces called c-Met. Normally, HGF fits into c-Met to activate a cascade of signals that promote cell growth, differentiation, and survival. Dihexa, despite being tiny compared to HGF, can attach to HGF in a way that “supercharges” its activity at the c-Met receptor. In essence, Dihexa acts like a molecular key that turns the lock of the c-Met receptor more efficiently. Structurally, by binding to HGF, Dihexa helps HGF form an active complex that triggers c-Met. This is remarkable – it means an engineered mini-peptide can hijack a major cell-growth pathway. The design of Dihexa took inspiration from naturally occurring peptides (like Pinealon or Testagen, which are tissue-derived regulators), yet Dihexa is fully synthetic and purpose-built. It’s an example of rational drug design merging with peptide biology: researchers identified a beneficial natural pathway (HGF/c-Met for neural growth) and crafted a peptide analog to mimic and enhance that pathway beyond what the body’s normal peptides do.
Physicochemically, Dihexa highlights the advantages of small peptides in therapeutics research. It has a low molecular weight and a structure stabilized against peptidases (the enzymes that typically digest peptides). Studies found that Dihexa is remarkably stable in the body of test animals, with a half-life on the order of days, not minutes. Its lipophilicity means it can travel through the normally restrictive blood–brain barrier, delivering its effects directly to the central nervous system. Unlike large protein growth factors, which might be too big or fragile to use in the brain, Dihexa’s compact form factor allows it to act as a surrogate neurotrophic signal. In summary, Dihexa’s molecular design bridges neurotrophic biology and synthetic chemistry: it is essentially a miniaturized, brain-penetrant growth factor mimetic. This exemplifies a new generation of peptides engineered to carry specific bioactivities – in Dihexa’s case, the ability to engage neural repair and connectivity pathways with precision.
Mechanistic Insights & Cellular Targets
How does Dihexa exert its effects on brain cells? Researchers have broken down its mechanism of action into several key themes:
- HGF/c-Met Pathway Activation: The cornerstone of Dihexa’s activity is its role as an HGF mimetic. Dihexa binds to hepatocyte growth factor and forms a complex that powerfully activates the c-Met receptor on cells. The c-Met pathway is normally involved in developmental growth and wound healing; in the brain, activating c-Met triggers signals that help neurons survive, grow new processes, and connect with each other. Preclinical studies show that Dihexa’s effects are dependent on this pathway: when c-Met is blocked or HGF is inhibited, Dihexa loses its procognitive benefits. By effectively turning on c-Met, Dihexa initiates a cascade (including PI3K/Akt and MAPK/ERK pathways) that promotes cell survival and growth. One result of this upstream activation is enhanced synaptogenesis – Dihexa, much like HGF itself, encourages neurons to form new synapses (connections) and dendritic spines (small protrusions where synapses form). This mechanism is distinct from typical neurotransmitter-based drugs; Dihexa isn’t simply boosting communication at existing synapses, it’s triggering the cell’s internal growth programs. Think of it as flipping a master switch that tells neurons to enter a pro-connectivity, pro-survival state.
- Synaptic Plasticity and Neurogenesis: Downstream of c-Met activation, Dihexa appears to bolster the fundamental processes of synaptic plasticity (the strengthening or creation of synapses) and potentially neurogenesis (the formation of new neurons) in certain brain regions. In lab experiments with brain slices and neuronal cultures, Dihexa treatment led to marked increases in long-term potentiation (LTP) – a cellular model of learning where synaptic connections become stronger. Additionally, researchers observed an increase in dendritic spine density on neurons exposed to Dihexa. Dendritic spines are tiny structures that house synapses; more spines generally indicate a greater capacity for connections and information storage. Not only did Dihexa create more spines, but the spines were larger and more mature (having wider “heads”), suggesting that the synapses were strong and functional. Supporting this, levels of key synaptic proteins were elevated – for instance, markers like PSD-95 and synapsin-1 (which are essential components of synapses) were present in the new connections, indicating they were proper, working synapses. In live animal studies, these cellular changes translated into better performance on memory tasks. Rodents treated with Dihexa showed improved learning and memory in mazes and object recognition tests, correlating with increased synaptic protein expression in the hippocampus (a brain region critical for memory). Molecular profiling from these models also hinted that Dihexa influences genes linked to plasticity: for example, brain-derived neurotrophic factor (BDNF) (a gene that supports neuron growth) and CREB (a protein that helps turn on memory-related genes) were modulated in response to Dihexa’s activation of growth pathways. An immediate-early gene called Arc, which is associated with LTP and memory consolidation, was also upregulated in some studies, reflecting heightened synaptic activity. All these changes paint a picture of Dihexa as a catalyst for a more connected and adaptable neural network, at least in the controlled settings of preclinical experiments.
- Cellular Protection and Mitochondrial Support: Beyond building new connections, Dihexa exhibits a profile of being neuroprotective. Neurons under stress (such as oxidative stress, toxin exposure, or metabolic impairment) benefit from the signals Dihexa initiates. In cell culture studies, neurons treated with Dihexa better survived challenges like excessive oxidative molecules or excitotoxic glutamate exposure, compared to untreated cells. One reason is that Dihexa, through c-Met activation, turns on pro-survival signaling cascades – notably the PI3K/Akt pathway and the ERK1/2 (MAPK) pathway, both of which are known to increase cell survival and bolster metabolism. As a result, Dihexa-treated cells maintain a healthier mitochondrial membrane potential and ATP production level (indicators that the cell’s “power plants,” the mitochondria, are functioning and intact). Concurrently, Dihexa seems to dial down the cellular pathways leading to apoptosis (programmed cell death). Researchers have noted reductions in caspase-3 activation (a key enzyme that executes cell death) and other apoptotic markers when Dihexa is present. In simpler terms, Dihexa sends a “stay alive and repair” signal to neurons: it not only helps them form new connections but also shields them from damage and energy loss. Some experiments even suggest that Dihexa can enhance the production of antioxidant defenses in neurons, meaning it might help neutralize harmful free radicals. Because of these protective effects, scientists have dubbed Dihexa a potential “neurorestorative signal amplifier” – it amplifies the brain’s own protective and restorative signals, preserving cells and their function in adverse conditions.
Preclinical Research Landscape
Extensive in vitro (cellular) and in vivo (animal) studies have been conducted to map out Dihexa’s effects. Below is an overview of what researchers have observed in the lab: In Vitro Studies In laboratory cell cultures, Dihexa has demonstrated a range of effects on neurons and supporting brain cells:
In laboratory cell cultures, Dihexa has demonstrated a range of effects on neurons and supporting brain cells:
- Neurite Outgrowth and Synaptic Marker Expression: When Dihexa is added to cultures of rat hippocampal neurons, the cells respond by growing more neurites – the extensions of neurons that develop into axons and dendrites. One study noted nearly a three-fold increase in dendritic spines (small protrusions where synapses form) on neurons treated with Dihexa compared to controls. These new spines weren’t empty stubs; they contained normal synaptic proteins, including the presynaptic protein synapsin-1 and the postsynaptic density protein PSD-95, indicating that real synapses were forming. The functional maturity of these synapses was confirmed by electrophysiological recordings showing increased AMPA receptor activity (a sign of active synaptic transmission) in Dihexa-treated neurons. This suggests Dihexa doesn’t just create cellular structures, it helps build working neural circuits in a dish.
- Increased Cell Survival Under Stress: Dihexa-treated neuronal cultures show greater resilience when exposed to various stressors. For example, in models of oxidative stress (which generates harmful reactive oxygen species) or excitotoxic stress (excessive stimulation by neurotransmitters), neurons pretreated with Dihexa have higher survival rates than untreated neurons. The peptide seems to pre-activate the cells’ defense mechanisms: Dihexa can elevate levels of antioxidant enzymes and protective proteins in cells, preparing them to withstand damage. Moreover, measurements of mitochondrial health (such as the maintenance of mitochondrial membrane potential) remain more stable in stressed neurons that received Dihexa. These findings align with the mechanistic insight that Dihexa triggers pro-survival signaling (Akt/ERK pathways), thereby guarding cells against injury.
- Activation of Growth Factor Signaling Cascades: On a molecular level, in vitro experiments confirm that Dihexa engages the HGF/c-Met pathway inside cells. When Dihexa is present (especially if a small amount of HGF is also present), researchers detect phosphorylation of c-Met – an activation mark on the receptor – along with downstream signaling events like Akt phosphorylation. If cells are treated with an HGF inhibitor or a c-Met blocker, those phosphorylation events – and Dihexa’s beneficial effects – are greatly reduced. This cause-and-effect relationship solidifies that Dihexa’s cellular actions are c-Met dependent. Additionally, gene expression assays in cultured neurons show that Dihexa can upregulate neurotrophic genes (such as BDNF) and immediate early genes linked to synaptic activity (Arc), consistent with the idea that it’s turning on a pro-plasticity genetic program.
- Dose-Dependent Responses without Hormonal Effects: Researchers have explored a range of Dihexa concentrations in vitro and found that its effects are dose-dependent within a physiological range – too low and there’s minimal impact, too high and the effects plateau or even diminish (a common “bell curve” response for growth factors). Importantly, Dihexa does not act like a hormone or traditional drug at the cellular level; it doesn’t indiscriminately stimulate activity. Instead, its impact is more modulatory: in healthy, unstressed neurons, adding Dihexa doesn’t make them hyperactive or cause abnormal firing. But in challenged or suboptimally functioning neurons, Dihexa provides a noticeable boost, restoring activity toward a normal baseline. This nuanced action – enhancing what’s needed without over-driving – is characteristic of many bioregulatory peptides. It underscores that Dihexa’s role in vitro is as a gene and growth modulator rather than a direct neurotransmitter agonist.
Animal Studies
Preclinical in vivo studies (primarily in rodents) have been pivotal in evaluating Dihexa’s effects on complex brain functions and injury models:
- Cognitive Impairment Models: In a hallmark study, Dihexa was tested in rats that had chemically induced cognitive deficits. These rats were given scopolamine, a compound that impairs learning and memory (mimicking certain Alzheimer’s-like cognitive symptoms). Dihexa treatment dramatically improved the rats’ performance in maze tests. Specifically, scopolamine-treated rats that received Dihexa were able to learn the location of a hidden platform in a water maze much faster than scopolamine-damaged rats without Dihexa. In fact, at optimal doses, the Dihexa-treated impaired rats performed almost as well as normal, healthy rats on this task. This indicates that Dihexa effectively reversed learning deficits in that model. Notably, when the same experiment was done in aged rats (natural cognitive decline model), orally delivered Dihexa also led to improvements in memory performance – though the effect was more modest due to variability in how much baseline impairment each old rat had. These results suggest that Dihexa can enhance cognitive function particularly when there is an underlying deficit, aligning with its role as a restorative agent.
- Neurodegenerative Disease Context: Although no human trials have been conducted, researchers have explored Dihexa in animal models relevant to neurodegenerative diseases. In rodent models mimicking aspects of Alzheimer’s disease, such as transgenic mice that accumulate amyloid beta or have memory impairments, Dihexa treatment led to improvements in memory tests and synaptic density. For instance, treated animals showed greater recognition of novel objects and improved spatial memory compared to untreated diseased mice, alongside biochemical signs of increased synaptic protein levels in the brain. These promising findings hint that Dihexa’s synaptogenic effects could counteract the synapse loss central to neurodegenerative disorders. Additionally, because Dihexa activates cell survival pathways, scientists have hypothesized it might protect neurons from degenerative processes – an idea supported by observations like reduced markers of inflammation and cell stress in Dihexa-treated AD-model mice.
- Traumatic Brain Injury and Neural Repair: An exciting area of ongoing research is whether Dihexa can aid recovery from brain injuries. Early preclinical experiments in models of traumatic brain injury (TBI) and stroke are examining Dihexa’s ability to spur regrowth of damaged neural circuits. Initial results are encouraging rodents with induced brain injuries that received Dihexa have shown better motor function recovery and cognitive outcomes than those without, suggesting the peptide helped the brain reorganize and heal. In these models, scientists observed signs of axonal sprouting (growth of new nerve fibers) and synaptic reconstruction in regions surrounding the injury. Dihexa’s activation of HGF/c-Met is a plausible mechanism here, as the HGF pathway is known to be involved in tissue repair and regeneration. By amplifying this pathway, Dihexa might create a more permissive environment for the brain to rewire after injury. It’s worth noting that this research is still in early stages – these studies aim to understand biological potential, not to declare a therapy. But they illustrate how Dihexa is being used as a tool to ask, “Can we improve the brain’s self-repair mechanisms after damage?”.
- Systems and Behavioral Outcomes: Across animal studies, Dihexa’s systemic effects appear consistent with its cellular actions. Treated animals often display not just improved test scores in mazes or memory challenges, but also underlying neural changes. Post-mortem analyses of rodent brains have revealed higher synapse counts and increased expression of neurotrophic factors in Dihexa groups. For example, the density of dendritic spines in the hippocampus was greater in Dihexa-treated rats, aligning with behavioral improvements. Importantly, these studies have also monitored safety signals: short-term dosing in animals did not produce obvious toxicity or organ damage, and notably, even though c-Met is an oncogene (a cancer-related gene), activating it with Dihexa did not show signs of inducing tumors in these short-term studies. Still, caution is the rule – researchers stress that positive results in rodents are no guarantee of success in humans, and much more work is needed to understand long-term effects. As of now, Dihexa’s preclinical profile is that of a potent cognitive enhancer and neural regenerator in the lab, giving scientists a powerful probe to study brain plasticity.
Conclusion
Dihexa stands out as an experimental peptide that illuminates how small molecules can profoundly influence brain biology. Through its unique action of modulating the HGF/c-Met growth factor system, Dihexa has demonstrated the ability to promote neuronal growth, strengthen synaptic connections, and support cell survival – all pivotal elements of brain plasticity and repair. Importantly, everything known about Dihexa so far comes from preclinical research: cell cultures and animal studies have been the exclusive arena for these discoveries. Dihexa is not approved for clinical use and remains a research tool.
Nonetheless, what scientists have learned from Dihexa carries significant implications. It represents a “model molecule” for understanding how we might awaken the brain’s latent regenerative capacities. The fact that a peptide so small can engage deep biological circuits of growth and regeneration is encouraging – it suggests that, in principle, neural circuits can be rebuilt or preserved by precisely targeted signals.
In summary, Dihexa has opened a new window into the interplay between peptide chemistry and neural resilience. It bridges gaps between disciplines: tying together insights from molecular pharmacology, neuroscience, and even epigenetics. By studying Dihexa, researchers are gaining clues about how the brain’s connectivity and cognitive functions might be maintained or restored through biochemical means. Looking forward, the ongoing research on Dihexa and its analogs will continue to shed light on the boundaries of neuroplasticity. Each experiment with this peptide refines our understanding of how neurons can recover and how memory and learning processes might be bolstered at the molecular level. While much remains to be explored – and caution is warranted before any translation beyond the lab – Dihexa’s story so far is a compelling illustration of science’s progress toward harnessing the brain’s own regenerative language. It underlines a hopeful vision: that one day, knowledge gained from molecules like Dihexa could inform new strategies to combat neurodegeneration, cognitive decline, and brain injuries, helping to keep our neural connections strong throughout life.